
The element boron (B, atomic number 5 and atomic mass 11) is right above aluminum (Al, atomic number - Brainly.com

Aluminium Or Aluminum Is A Chemical Element In The Boron Group With Symbol Al And Atomic Number 13 Stock Photo, Picture And Royalty Free Image. Image 136730082.

An element 'X' has atomic number 13:; (a) Write its electron configuration.; (b) state the group to - Brainly.in

An element has atomic number 13 and an atomic mass of 27 (a) How many electrons are there in each atom of the
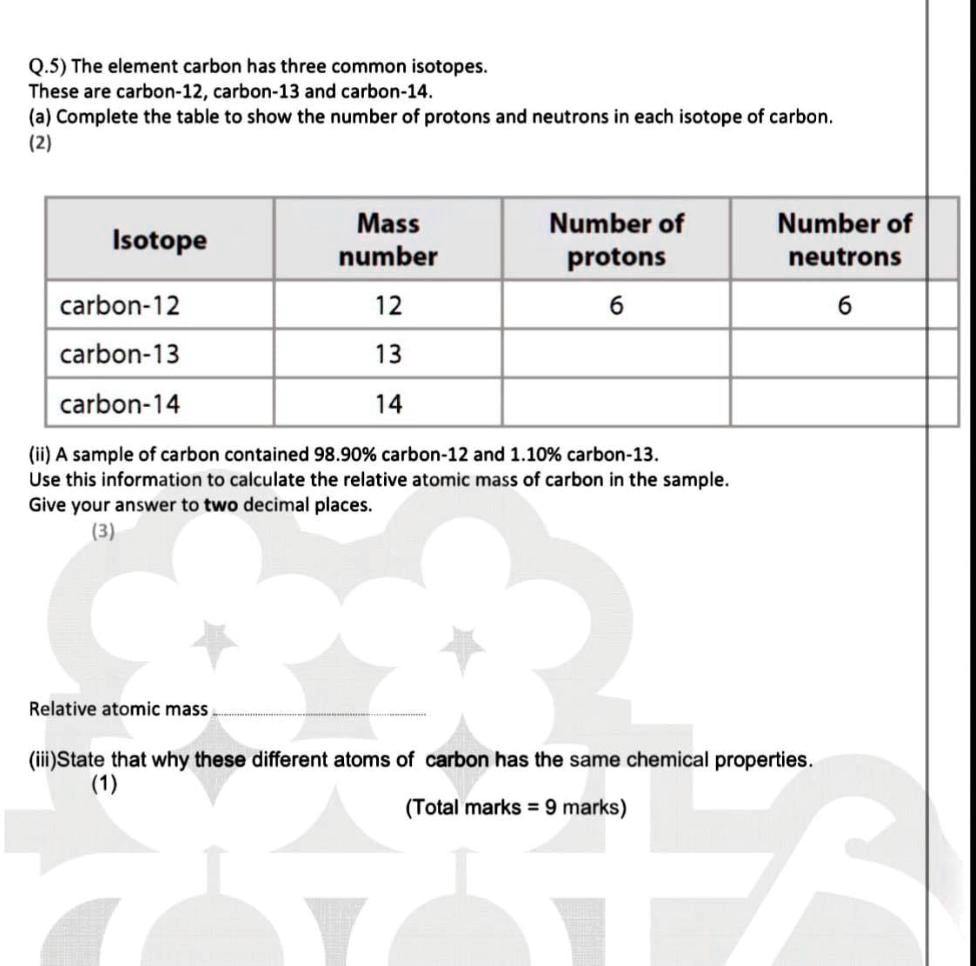
SOLVED:Q.5) The element carbon has three common isotopes: These are carbon-12, carbon-13 and carbon-14 (a) Complete the table to show the number of protons and neutrons in each isotope of carbon: (2)

Aluminum Is A Chemical Element With The Symbol Al And Atomic Number 13 With A Mass Of 27 U Aluminum Stone For Industrial Use Silver Ore Stock Photo - Download Image Now - iStock

The Periodic Table: atom, chemistry, dmitri, dobereiner, element, eng, mendeleev, newlands, periodic, set | Glogster EDU - Interactive multimedia posters

Write down the characteristics of the elements having atomic number 17.Electronic configuration Period number Group number Element family No. of valence electrons Valency Metal or non - metal .

SOLVED:A substance has 13 protons, 14 neulrons_ and 13 clectrons_ Which isotope is it? Al-13 B) Al-14 Al-27 D) Al-40 One isotope of oxygen has the atomic number 8 and the mass







/scientist-wearing-surgical-gloves-holding-piece-of-aluminium-scrap-in-aluminium-recycling-plant--close-up-180404744-5a4bde94aad52b00365fc147.jpg)
/what-are-the-first-20-elements-608820-FINAL-5b758ab446e0fb002c67279a.png)