
Solubility of PuO 2 (am,hyd) and the Formation of Pu(IV) Carbonate Complexes in Carbonate Solutions Containing 0.1–5.0 mol·dm −3 NaNO 3 | SpringerLink

AqSolDB, a curated reference set of aqueous solubility and 2D descriptors for a diverse set of compounds | Scientific Data
![What is the pH of a saturated solution of X(OH)3 will be if its solubility product = 2.7 × 10^-43 ?[Given: log 3 = 0.5, log 2 = 0.3] What is the pH of a saturated solution of X(OH)3 will be if its solubility product = 2.7 × 10^-43 ?[Given: log 3 = 0.5, log 2 = 0.3]](https://d2rrqu68q7r435.cloudfront.net/images/7984587/97259092-21e7-413a-aa0c-7fee11193a62.jpg)
What is the pH of a saturated solution of X(OH)3 will be if its solubility product = 2.7 × 10^-43 ?[Given: log 3 = 0.5, log 2 = 0.3]
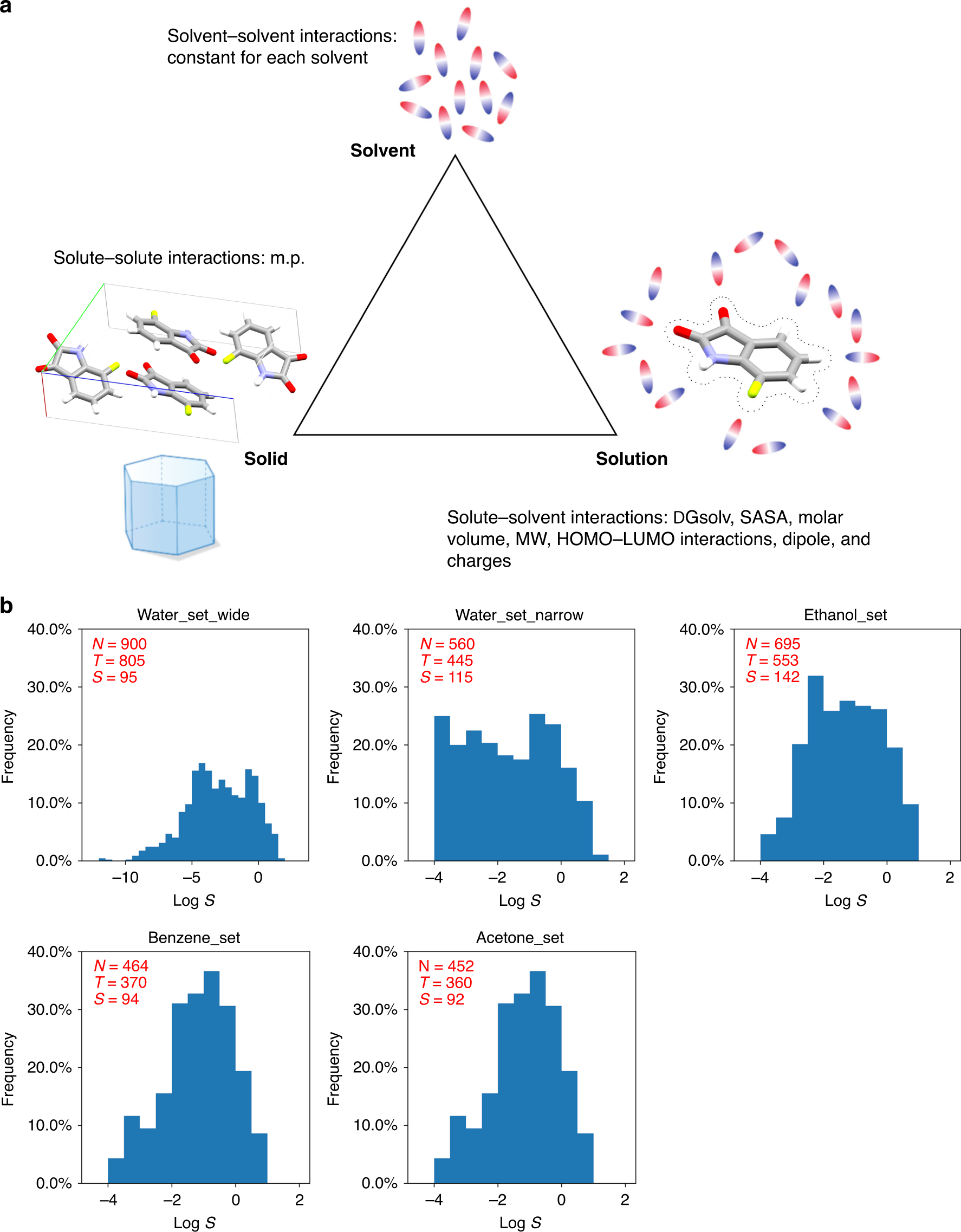
Machine learning with physicochemical relationships: solubility prediction in organic solvents and water | Nature Communications
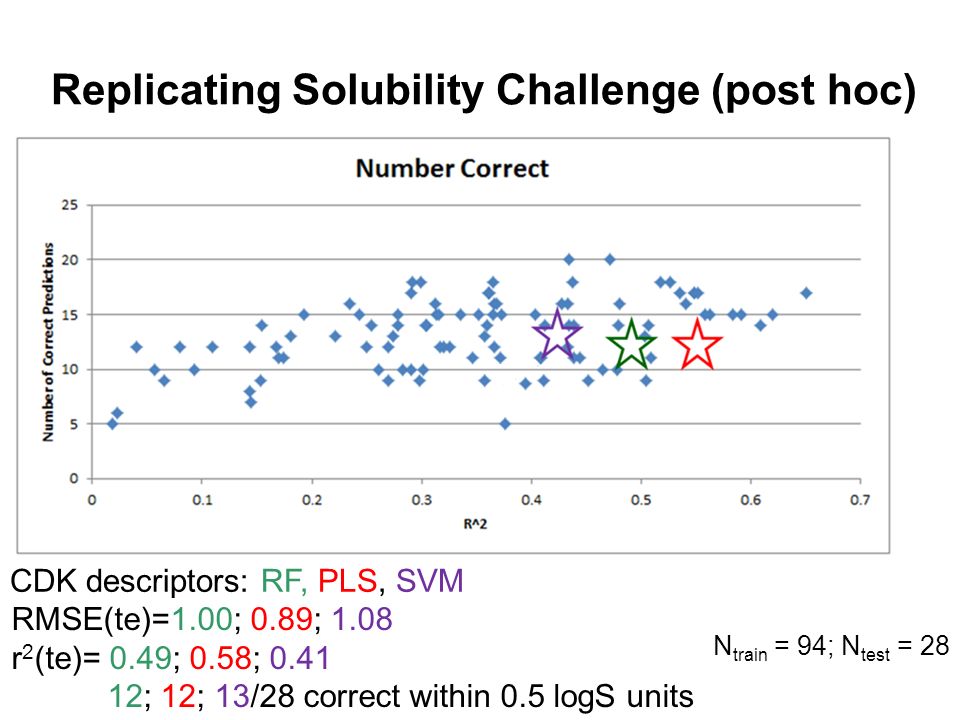
Quantum Chemical and Machine Learning Calculations of the Intrinsic Aqueous Solubility of Druglike Molecules Dr John Mitchell University of St Andrews. - ppt download

Figure 4 | Solubility of Zr(OH) 4 (am) and the Formation of Zr(IV) Carbonate Complexes in Carbonate Solutions Containing 0.1–5.0 mol·dm −3 NaNO 3 | SpringerLink
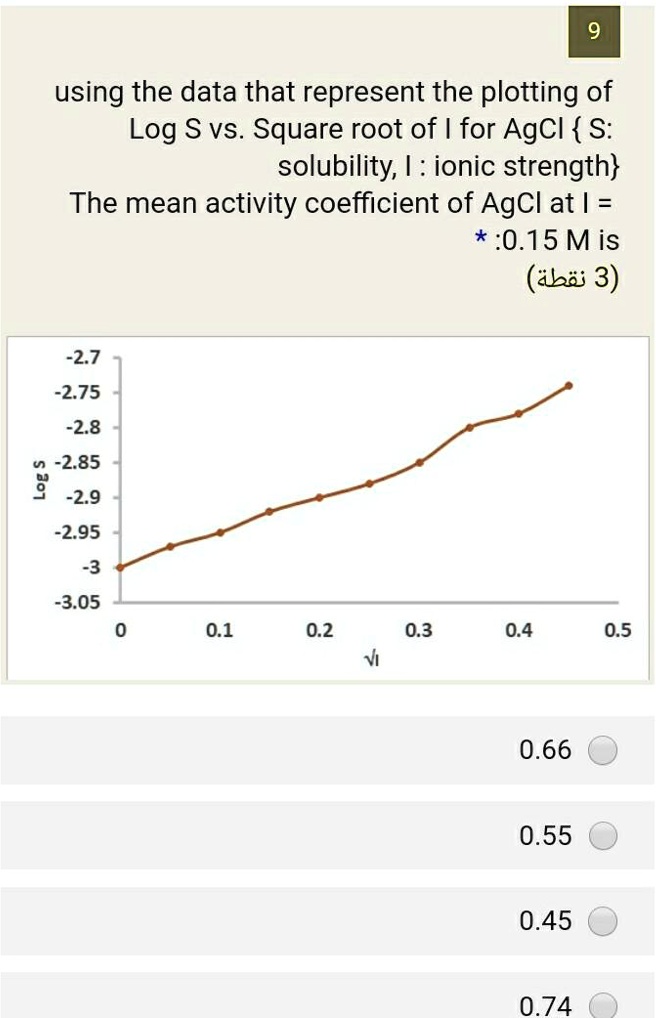

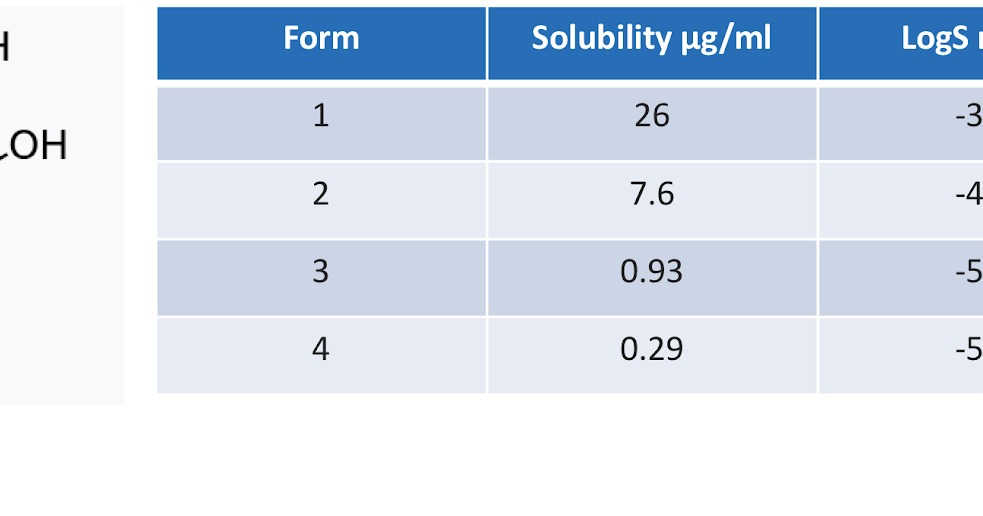




![Solubility of Zr(IV) in 0.5 mol·dm⁻³ NaNO3 as a function of [C]tot... | Download Scientific Diagram Solubility of Zr(IV) in 0.5 mol·dm⁻³ NaNO3 as a function of [C]tot... | Download Scientific Diagram](https://www.researchgate.net/publication/315922876/figure/fig2/AS:961857066131456@1606336003242/Solubility-of-ZrIV-in-05moldm-NaNO3-as-a-function-of-Ctot-after-ultrafiltration.gif)






