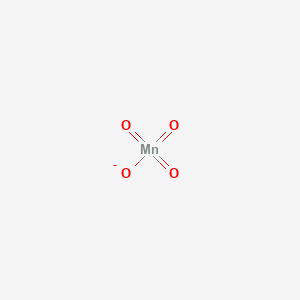
1a) Calculate the oxidation number for one atom of Mn in the KMnO4 using the reaction:... - HomeworkLib

SOLVED:An acidic solution of permanganate (MnO4" ions can oxidize nitrous acid t0 nitrate ions_ The products include dissolved Mn?+ ions Write the chemical equations describing the oxidation of nitrous acid and the

If S + O2→ SO2, H = - 298.2 kJ mole^-1 SO2 + 1/2 O2→ SO3, H = - 98.7 kJ mole^-1 SO3 + H2O → H2SO4, H = - 130.2 kJ
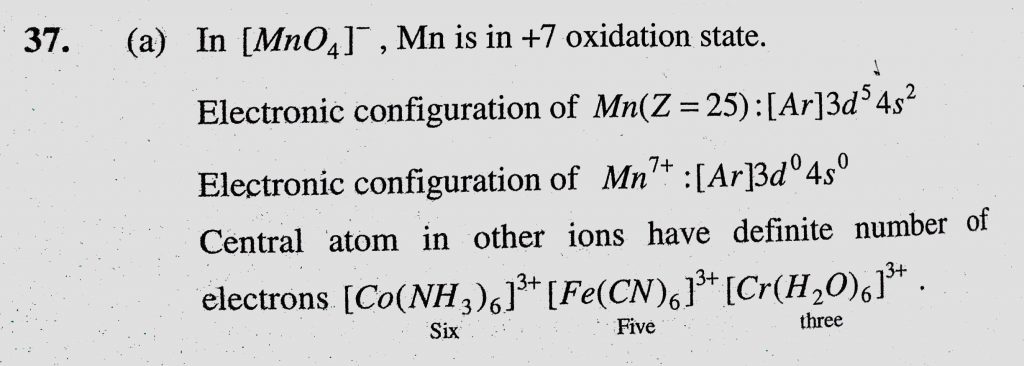
The complex ion which has no 'd' electrons in the central metal atom is (a) (MnO4)- (b) (Co(NH3)6) 3+ - Sahay Sir

Permanganate Anion, Chemical Structure. 3D Rendering. Atoms are Represented As Spheres with Conventional Color Coding: Manganese Stock Illustration - Illustration of oxygen, model: 188435872


![MnO4]2- - Manganate MnO4]2- - Manganate](https://www.chemtube3d.com/images/gallery/inorganicsjpgs/MnO42-.jpg)
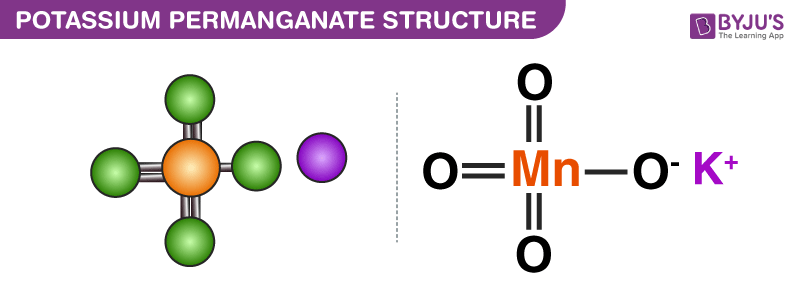



.PNG)